Published online 2015 Dec 2. doi: 10.1002/acn3.269
PMID: 26783551
The calculator has been adapted to estimate the Glasgow verbal score from the Glasgow eye and motor scores in intubated patients. There is a Paediatric Glasgow Coma Scale applicable to infants too young to speak - and the equivalent infant responses are given in the various sections below. This section discusses the Glasgow Coma Scale. Click on the link to find out more information about the Ranchos Los Amigos Scale. The Glasgow Coma Scale is based on a 15-point scale for estimating and categorizing the outcomes of brain injury on the basis of overall social capability or dependence on others.
This article has been cited by other articles in PMC.
Associated Data
Table S1. Consciousness development, prognosis factors, progress, and complications of J. W.'s first 8 months after the event. GCS, Glasgow Coma Scale; UWS, unresponsive wakefulness syndrome; MCS, minimally consciousness state; SSEP, somatosensory event‐related potentials; MRI, magnetic resonance imaging; MRSA, methicillin‐resistant Staphylococcus aureus; ERP, event‐related potentials.
GUID: 9C37929D-26AE-4343-A585-A6493787D73C
Abstract
To demonstrate the possibility for hidden rehabilitation potential even following most severe brain injury and the uncertainty of current prognosis factors for coma and unresponsive wakefulness syndrome, we detail the rehabilitation of J. W., after coma from traumatic brain injury. Originally, with many negative prognosis factors and several medical complications, prognosis was devastating. But, with continuing treatment, J. W. improved to a high level of independence in everyday life. This shows the need for rehabilitation research to further specify the “prognostic power” of various combinations of prognosis factors, so that practitioners can come to accurate single‐case recommendations when both positive and negative predictors are present.
Introduction
Severe traumatic brain injury (TBI) often results in coma and subsequent disorders of consciousness such as unresponsive wakefulness syndrome (UWS) (former vegetative state)1, 2, 3 or minimally conscious state (MCS).4 The survival rate after a TBI, severe enough to cause deep coma and low Glasgow Coma Scale (GCS) scores, is generally poor, even in young adults. Studies show a very high overall mortality, ranging between 76% and 89%.5, 6, 7 Of the surviving patients, only very few recover to a good outcome. The majority of the survivors do so with permanent disorders of consciousness or severe disabilities (see Table 1). In acute coma, most prognosis factors are essentially able to predict a negative outcome. Two powerful predictors, usually obtained right at the accident site, are the patient's pupillary reactions and GCS scores. Various studies report significantly increased mortality for patients with initial GCS scores of ≤5 compared to those with higher GCS scores.5, 6, 7, 8, 9 In fact, the chances of survival with a GCS less than 5 are considered so poor, that in regions with limited resources, patients with a GCS of ≤5 are not even admitted to intensive care units (ICU) (as reported in Jain et al.5).
Table 1
Prognosis factors for coma patients with severe TBI
| References | N/Age group | Type of traumatic brain injury | Initial GCS score | Overall mortality | Mortality with fixed pupils | Mortality with low GCS plus fixed pupils | Good outcome |
|---|---|---|---|---|---|---|---|
| Jain et al.5 | 102/6 to 75 years | Isolated blunt head injury | 3–5 | 76.5% | 83.1% | 10 patients (GOS 4 and 5) | |
| Demetriades et al.6 | 760/from under 20 to over 55 years | Blunt and penetrating trauma | 3 | 76% | 177 survivors, 18 with preinjury functional capacity | ||
| Kotwica and Jokubowski7 | 111/18–82 years | Blunt and penetrating trauma | 3 | 89% | 2 patients GOS 4 2 patients GOS 5 | ||
| White et al.9 | 136/0–17 years | Closed head injury | ≤8 | 24% | 93.75% of those with GCS under 5 | ||
| Lieberman et al.11 | 137/≥14 years | Blunt and penetrating trauma | 3 | 92% | N = 104 100% died | 11 survivors 1 patient with FIM 16 3 patients with FIM 20 | |
| Rovalis and Kotsou10 | 345/16–70 years | Closed head injuries | ≤8 | N = 119, GCS 3–5, 96.6% died | 151 favorable outcomes, not further specified | ||
| Sigorini et al.8 | 372/≥14 years | Blunt and penetrating trauma | ≤15 | 23% | 62% | 279 survivors, outcome not further specified |
TBI, traumatic brain injuries; GCS, Glasgow Coma Scale; GOS, Glasgow Outcome Scale (1 = dead, 2 = UWS, 3 = severe disability, completely dependent, 4 = moderate disability, employment possible with special equipment, 5 = low disability, minor neurological and psychological deficits); FIM, functional independence measure (18 items from which 13 are motor tasks and 5 cognitive tasks. Higher scores indicate higher levels of function).
Absent pupillary reactions are also very powerful negative predictors. Again, Jain and colleagues found that poor pupillary reactions increase the odds ratio for death to 5.5.5 A combination of both, fixed pupils and a low GCS has even been found to be associated with nearly absolute mortality (96.6%10 and 100%11). Other negative predictors (indicating death or long‐lasting UWS) obtained within the first days after the accident are the presence of hematoma on computer tomograms (CT), an age over 55 years, clinical complications (like fever or seizures), and the absence of any motor response to painful stimulation.8, 12, 13, 14
Once a TBI‐related UWS is reached, in children, overall mortality drops (14%) but chances of an unfavorable outcome (long‐lasting UWS or severe disabilities) are still high (68%). For adults, an unfavorable outcome (death or long‐lasting UWS) of 48% is reported, only 7% reaching a good outcome.2, 3 Higashi et al. even report a mortality of 66% and only three patients (of the 110) were able to communicate at a 5‐year follow‐up.15
In general, a more positive prognosis is made for patients with rapid signs of recovery (within the first days and weeks) and less than 40 years of age.2, 3 A more specific positive prognosis factor is an EEG reactivity to external stimulation.16, 17 Such reactivity has been shown to be lacking in 92% of all patients with severe EEG slowing (<4 Hz). None of the patients with such slow background EEG showed any improvement.16 For MCS, the same predictors are assumed, although there are studies that suggest that the outcome of MCS patients cannot be foretold by UWS predictors.4, 18
All predictors have been scientifically established for groups of patients. However, clinical prognoses for single patients and clinical counseling of family members are also routinely based on these factors.
Here, we illustrate the uncertainty of such population‐based predictions by presenting a case of good recovery despite a range of negative predictors which made even survival very unlikely.
Case Description
Patient J. W. experienced a serious TBI due to a car accident at the age of 18 years. At the accident site, he presented a GCS score of 4, wide, fixed pupils, and decerebrate rigidity of all four extremities. The patient was hospitalized at the ICU “Universitätsklinikum Würzburg,” Germany. Computer tomography (CT) scans revealed diffuse brain edema and subdural bleeding with midline shift (see Fig. Fig.1A).1A). An emergency decompression craniotomy was performed to reduce intracranial pressure by removing parts of the cranium.19 After operation, J. W. still displayed fixed pupils, no corneal reflex, and no motor response to pain. He further displayed seizures affecting the left side of his face. The family was told that brain damage was devastating and that survival was very unlikely. But the family decided to continue life support. J. W. remained in the ICU for 12 days after which he was transferred to a neurological rehabilitation facility, “Kliniken Schmieder,” Allensbach, Germany. At admission he displayed:
- Coma
- Acute subdural hematoma, right side
- Brain swelling
- Artificial respiration
- Abnormal pupillary functions
- Severe spastic tetraplegia
- Tracheal cannula
- Percutaneous gastrostomy
- Decerebrate rigidity of all extremities
(A) Computed tomography (CT) from the day of the accident showing acute right side hematoma with midline shift. (B) CT from 5 months after the event after cranium reimplantation showing an epidural hematoma, which required a second surgery. (C) Event‐related potentials from an oddball paradigm at electrode Cz. Identifiable are congruent but slightly delayed N100 responses for frequent and novel tones with a peak latency of 128 msec. A discernible but delayed P300 with a peak latency at 457 msec is displayed for novels, indicating the detection and intensified processing of new and unexpected events. (D) Event‐related potentials from a N400 paradigm at electrode C4. Identifiable N100, more pronounced for semantically correct sentence endings and smaller for incorrect sentence endings. Between 400 and 800 msec, a clearly visible but delayed N400 is displayed, indicating the detection and intensified processing of semantic violations.
Rehabilitation course with all positive and negative predictors is also detailed in Table S1. During the first month in rehabilitation, the patient was still on ventilation and autonomically unstable with sweating, tachycardia, and hypertension. No change in mental status was observed.
In the second month, the EEG showed severe slowing, with delta–theta dominance. Magnetic resonance imaging (MRI) displayed a right‐frontal subdural hematoma and an asymmetric, right dominant ventricle system enlargement without midline displacement, as well as parenchyma lesions with gliosis in temporal regions and in the white matter surrounding the posterior horn of the lateral ventricle. Still, by the end of the second month, J. W. could be weaned from ventilation, although there was no change in cognitive functions.
By the third month, the patient reached the status of MCS. However, there were no vocalizations other than groaning to painful stimulation. No communication channel and no command following could be established.
After 4 months, a newly conducted MRI revealed considerable brain atrophy, multiple parenchyma lesions, diffuse shear lesions, but no periventricular transudation of cerebrospinal fluid.
Five months after the event the cranium reimplantation was scheduled. After the operation, various complications occurred, including an epidural hematoma, which required a new surgery (see CT in Fig. Fig.1B).1B). No clinical improvement was apparent.
After 7 months, J. W. began with situation‐independent screaming. This lasted for over 4 weeks. He screamed and moaned during his “wake” phases, which did not change with painkillers or other medication efforts. However, J. W.'s EEG now showed an almost normal alpha rhythm. Event‐related potentials (ERPs) for tone discrimination (P300) and semantic speech processing (N400) were clearly identifiable, albeit latency delayed (see Fig. Fig.1C1C and D).
Eight months after the event, J. W. began to close his eyes on command, clinically indicating progress to MCS+. Nine months after the event, the patient established a communication channel, first via eye blink, and shortly thereafter also via nods and head shaking. Communication is described as increasingly adequate which implies that the patient improved beyond MCS. At this point, after 9 months of intensive treatment, he was sent home from early rehabilitation.
At home, J. W. continued on an intensive therapy program: 2 h of occupational therapy, 3 h of physiotherapy, and 2 h of speech therapy a week. He regained better body control, so that after 6 months at home, the patient was able to walk alone for about 20 m on flat ground albeit walking was still very ataxic and unstable. He also regained speech and was able to communicate adequately, although with aphasic symptoms. Epileptic seizures continued to occur about once a month.
After 6 months at home, the patient was rehospitalized for 10 weeks in the rehabilitation facility (Kliniken Schmieder, Germany) to improve his gait, his memory capacity, and general independence. During this time, one and a half years after the car accident, J. W. underwent a neuropsychological evaluation. He was fully oriented, displayed normal short‐term memory, normal verbal intelligence, and average executive performance. He still showed weaknesses in reproduction of geometrical figures and text information, below‐average long‐term memory capacity, and generally slowed information processing. He continued his therapy schedule in reduced form after rehabilitation for years after the accident.
Now, 10 years after the event, J. W. is able to walk independently, use stairs, and even ride his bicycle again. He has reached independence in all activities of daily living. His memory functions are still reduced but he has learned to cope. His speech is slow but his vocabulary and intonation are perfectly normal. He has communicated with the researchers personally, via telephone and e‐mail. J. W. is employed and lives close by, but independently, from his parents.
Discussion
This case illustrates three points. First, good recovery is possible despite a range of powerful negative predictors (low GCS score, fixed pupils, abnormal motor findings, various medical complications, and epileptic seizures). It should be kept in mind that every medical prognostic test is associated with “false negatives.” The prediction errors may be small for certain prognosis factors (like low GCS scores in combination with fixed pupils10, 11), but they are almost never 0. Even with error rates estimated to be 0%, there are usually non‐zero confidence intervals (CI) due to small sample sizes. The study of Lieberman and colleagues illustrates that fact with 104 patients with low GCS and fixed pupils: Although nobody survived, CI range was from 0.00 to 3.38.11 The case of J. W. highlights that very point.
Second, little is known about the interaction of positive and negative predictors. Was it only the patient's youth that outweighed all the other seemingly devastating negative predictors? Recently, attempts have been made to develop “multifactor models” for acute coma outcome. So far, these models take very different predictors into account and reach considerably different conclusions. For example, Jain et al.5 include three factors, namely the patient's pupil response, whether the patient needs ventilation right after the event, and whether the GCS improves by at least two points within the first 24 h. The IMPACT project from 201020 on the other hand, takes age, motor score, pupils, cause, CT findings, and laboratory results into account. Retrospectively inserting J. W.'s data result in a 15% survival chance with no further specification of outcome in Jain's model, whereas IMPACT forecasts a 35% chance of favorable outcome (better than UWS). Clearly, future studies should further address the question of “prognosis profiles” of single patients and should also specify outcome levels in greater detail. This would help practitioners to come to accurate conclusions in single cases where positive and negative predictors are present at the same time.
Finally, this case illustrates the value of multimodal intensive early and late rehabilitation, as well as the usefulness of continued treatment at home and interval rehospitalization. Continuous therapy helped the patient to improve over a period of 10 years, so that now an independent life is possible.
Authors' Contribution
I. S.: acquisition of medical information, analysis, and interpretation of ERP data and drafting the manuscript. M. K.: acquisition of medical information and revision of the manuscript. J. K.: drafting and revising the manuscript.
Supporting information
Table S1. Consciousness development, prognosis factors, progress, and complications of J. W.'s first 8 months after the event. GCS, Glasgow Coma Scale; UWS, unresponsive wakefulness syndrome; MCS, minimally consciousness state; SSEP, somatosensory event‐related potentials; MRI, magnetic resonance imaging; MRSA, methicillin‐resistant Staphylococcus aureus; ERP, event‐related potentials.
Acknowledgments
We thank the patient and the patient's family for their cooperation and contributions to this study. We also thank the clinics for insights into medical documentation. This study was funded by the state of Baden‐Württembergs ministry of science, research and the arts and the DFG Cluster of Excellence Cognitive Interaction Technology (CITEC).
References
1. Laureys S, Celesia GG, Cohadon F, et al. Unresponsive wakefulness syndrome: a new name for the vegetative state or apallic syndrome. BMC Med2010;8:68. [PMC free article] [PubMed] [Google Scholar]
2. Medical aspects of the persistent vegetative state (1). The Multi‐Society Task Force on PVS. N Engl J Med1994;330:1499–1508. [PubMed] [Google Scholar]
3. Medical aspects of the persistent vegetative state (2). The Multi‐Society Task Force on PVS. N Engl J Med1994;330:1572–1579. [PubMed] [Google Scholar]
4. Giacino JT, Ashwal S, Childs N, et al. The minimally conscious state: definition and diagnostic criteria. Neurology2002;58:349–353. [PubMed] [Google Scholar]
5. Jain S, Dharap SB, Gore MA. Early prediction of outcome in very severe closed head injury. Injury2008;39:598–603. [PubMed] [Google Scholar]
6. Demetriades D, Kuncir E, Velmahos GC, et al. Outcome and prognostic factors in head injuries with an admission Glasgow Coma Scale score of 3. Arch Surg2004;139:1066–1068. [PubMed] [Google Scholar]
7. Kotwica Z, Jakubowski JK. Head‐injured adult patients with GCS of 3 on admission–who have a chance to survive?Acta Neurochir1995;133:56–59. [PubMed] [Google Scholar]
8. Signorini DF, Andrews PJ, Jones PA, et al. Predicting survival using simple clinical variables: a case study in traumatic brain injury. J Neurol Neurosurg Psychiatry1999;66:20–25. [PMC free article] [PubMed] [Google Scholar]
9. White JR, Farukhi Z, Bull C, et al. Predictors of outcome in severely head‐injured children. Crit Care Med2001;29:534–540. [PubMed] [Google Scholar]
10. Rovlias A, Kotsou S. Classification and regression tree for prediction of outcome after severe head injury using simple clinical and laboratory variables. J Neurotrauma2004;21:886–893. [PubMed] [Google Scholar]
11. Lieberman JD, Pasquale MD, Garcia R, et al. Use of admission Glasgow Coma Score, pupil size, and pupil reactivity to determine outcome for trauma patients. J Trauma2003;55:437–442; discussion 442–3. [PubMed] [Google Scholar]
12. Acker SN, Ross JT, Partrick DA, et al. Glasgow motor scale alone is equivalent to Glasgow Coma Scale at identifying children at risk for serious traumatic brain injury. J Trauma Acute Care Surg2014;77:304–309. [PubMed] [Google Scholar]
13. Cook RJ, Fearnside MR, McDougall P, et al. The Westmead head injury project: outcome prediction in acute subdural haematoma. J Clin Neurosci1996;3:143–148. [PubMed] [Google Scholar]
14. Fearnside MR, Cook RJ, McDougall P, et al. The Westmead Head Injury Project outcome in severe head injury. A comparative analysis of pre‐hospital, clinical and CT variables. Br J Neurosurg1993;7:267–279. [PubMed] [Google Scholar]
15. Higashi K, Hatano M, Abiko S, et al. Five‐year follow‐up study of patients with persistent vegetative state. J Neurol Neurosurg Psychiatry1981;44:552–554. [PMC free article] [PubMed] [Google Scholar]
16. Kotchoubey B, Lang S, Mezger G, et al. Information processing in severe disorders of consciousness: vegetative state and minimally conscious state. Clin Neurophysiol2005;116:2441–2453. [PubMed] [Google Scholar]
17. Steppacher I, Eickhoff S, Jordanov T, et al. N400 predicts recovery from disorders of consciousness. Ann Neurol2013;73:594–602. DOI: 10.1002/ana.23835. [PubMed] [Google Scholar]
18. Steppacher I, Kaps M, Kissler J. Will time heal? A long‐term follow‐up of severe disorders of consciousness. Ann Clin Transl Neurol2014;1:401–408. [PMC free article] [PubMed] [Google Scholar]
19. Pechmann A, Anastasopoulos C, Korinthenberg R, et al. Decompressive craniectomy after severe traumatic brain injury in children: complications and outcome. Neuropediatrics2015;46:5–12. [PubMed] [Google Scholar]
20. Maas AI, Steyerberg EW, Marmarou A, et al. IMPACT recommendations for improving the design and analysis of clinical trials in moderate to severe traumatic brain injury. Neurotherapeutics2010;2010:127–134. [PMC free article] [PubMed] [Google Scholar]
Articles from Annals of Clinical and Translational Neurology are provided here courtesy of Wiley-Blackwell
| Glasgow Coma Scale | |
|---|---|
| Medical diagnostics | |
| MeSH | D015600 |
| LOINC | 35088-4 |
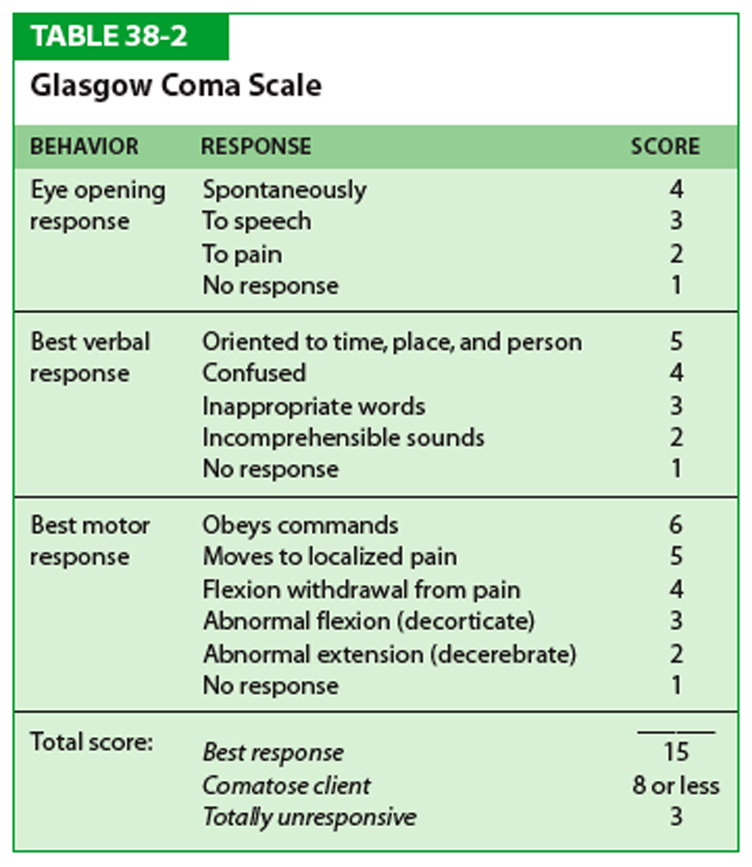
The Glasgow Coma Scale (GCS) is a neurologicalscale which aims to give a reliable and objective way of recording the state of a person's consciousness for initial as well as subsequent assessment. A person is assessed against the criteria of the scale, and the resulting points give a person's score between 3 (indicating deep unconsciousness) and either 14 (original scale) or 15 (more widely used modified or revised scale).
GCS was initially used to assess a person's level of consciousness after a head injury, and the scale is now used by emergency medical services, nurses, and physicians as being applicable to all acute medical and trauma patients. In hospitals, it is also used in monitoring patients in intensive care units.
The scale was published in 1974 by Graham Teasdale and Bryan J. Jennett, both professors of neurosurgery at the University of Glasgow's Institute of Neurological Sciences at the city's Southern General Hospital.
GCS is used as part of several ICU scoring systems, including APACHE II, SAPS II, and SOFA, to assess the status of the central nervous system. The initial indication for use of the GCS was serial assessments of people with traumatic brain injury[1] and coma for at least six hours in the neurosurgical ICU setting, though it is commonly used throughout hospital departments. The similar Rancho Los Amigos Scale, is used to assess the recovery of traumatic brain injury.
GCS was updated following a review of the helpfulness and usefulness of the scale from clinicians. It was decided that several things required updating, like the Eye Response element, meaning that instead of responding to 'Painful Stimuli' being regarded as a 2, a person that opens their eyes in response to pressure is now considered a 2 in the Eye Response element.[2]
- 1Elements of the scale
- 6References
Elements of the scale[edit]
| 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|
| Eye | Does not open eyes | Opens eyes in response to pain | Opens eyes in response to voice | Opens eyes spontaneously | N/A | N/A |
| Verbal | Makes no sounds | Makes sounds | Words | Confused, disoriented | Oriented, converses normally | N/A |
| Motor | Makes no movements | Extension to painful stimuli (decerebrate response) | Abnormal flexion to painful stimuli (decorticate response) | Flexion / Withdrawal to painful stimuli | Localizes to painful stimuli | Obeys commands |
Note that a motor response in any limb is acceptable.[4]The scale is composed of three tests: eye, verbal and motor responses. The three values separately as well as their sum are considered. The lowest possible GCS (graded 1 in each element) is 3 (deep coma or death), while the highest is 15 (fully awake person).
Eye response (E)[edit]
There are four grades starting with the most severe:
- No opening of the eye
- Eye opening in response to pain stimulus. (a peripheral pain stimulus, such as squeezing the lunula area of the person's fingernail is more effective than a central stimulus such as a trapezius squeeze, due to a grimacing effect).[5]
- Eye opening to speech. (Not to be confused with the awakening of a sleeping person; such people receive a score of 4, not 3.)
- Eyes opening spontaneously
Verbal response (V)[edit]
There are five grades starting with the most severe:
- No verbal response
- Incomprehensible sounds. (Moaning but no words.)
- Inappropriate words. (Random or exclamatory articulated speech, but no conversational exchange. Speaks words but no sentences.)
- Confused. (The person responds to questions coherently but there is some disorientation and confusion.)
- Oriented. (Person responds coherently and appropriately to questions such as the person’s name and age, where they are and why, the year, month, etc.)
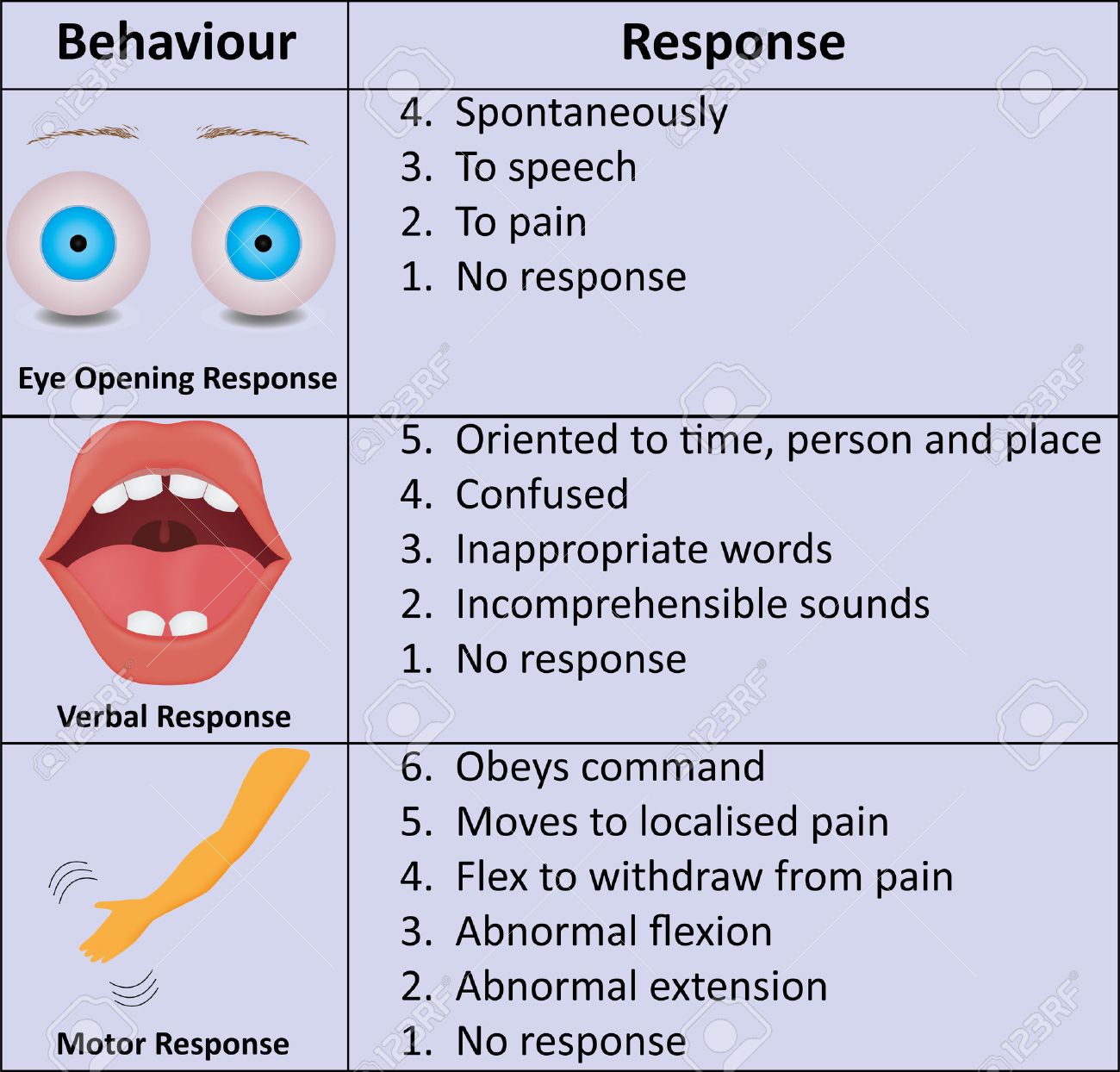
Motor response (M)[edit]
There are six grades:
- No motor response
- Decerebrate posturing accentuated by pain (extensor response: adduction of arm, internal rotation of shoulder, pronation of forearm and extension at elbow, flexion of wrist and fingers, leg extension, plantarflexion of foot)
- Decorticate posturing accentuated by pain (flexor response: internal rotation of shoulder, flexion of forearm and wrist with clenched fist, leg extension, plantarflexion of foot)
- Withdrawal from pain (absence of abnormal posturing; unable to lift hand past chin with supraorbital pain but does pull away when nailbed is pinched)
- Localizes to pain (purposeful movements towards painful stimuli; e.g., brings hand up beyond chin when supraorbital pressure applied)
- Obeys commands (the person does simple things as asked)
Interpretation[edit]
Individual elements as well as the sum of the score are important. Hence, the score is expressed in the form 'GCS 9 = E2 V4 M3 at 07:35'.
Generally, brain injury is classified as:
- Severe, GCS < 8–9
- Moderate, GCS 8 or 9–12 (controversial)[6]
- Minor, GCS ≥ 13.
Tracheal intubation and severe facial/eye swelling or damage make it impossible to test the verbal and eye responses. In these circumstances, the score is given as 1 with a modifier attached (e.g. 'E1c', where 'c' = closed, or 'V1t' where t = tube). Often the 1 is left out, so the scale reads Ec or Vt. A composite might be 'GCS 5tc'. This would mean, for example, eyes closed because of swelling = 1, intubated = 1, leaving a motor score of 3 for 'abnormal flexion'.
The GCS has limited applicability to children, especially below the age of 36 months (where the verbal performance of even a healthy child would be expected to be poor). Consequently, the Paediatric Glasgow Coma Scale was developed for assessing younger children.
Revisions[edit]
- Glasgow Coma Scale: While the 15-point scale is the predominant one in use, this is in fact a modification and is more correctly referred to as the Modified Glasgow Coma Scale. The original scale was a 14-point scale, omitting the category of 'abnormal flexion'. Some centres still use this older scale, but most (including the Glasgow unit where the original work was done) have adopted the modified one.
- The Rappaport Coma/Near Coma Scale made other changes.
- Meredith W., Rutledge R, Fakhry SM, EMery S, Kromhout-Schiro S have proposed calculating the verbal score based on the measurable eye and motor responses.
- The most widespread revision has been the Simplified Motor and Verbal Scales which shorten the respective sections of the GCS without loss of accuracy.[7]
- The GCS for intubated people is scored out of 10 as the verbal component falls away
Controversy[edit]
The GCS has come under pressure from some researchers who take issue with the scale's poor inter-rater reliability and lack of prognostic utility.[8] Although there is no agreed-upon alternative, newer scores such as the Simplified motor scale and FOUR score have also been developed as improvements to the GCS.[9] Although the inter-rater reliability of these newer scores has been slightly higher than that of the GCS, they have not gained consensus as replacements.[10]
See also[edit]
- AVPU scale
References[edit]
- ^Teasdale G, Jennett B (1974). 'Assessment of coma and impaired consciousness. A practical scale'. Lancet. 2 (7872): 81–4. doi:10.1016/S0140-6736(74)91639-0. PMID4136544.
- ^'What's new - Glasgow Coma Scale'. www.glasgowcomascale.org. Retrieved 2018-06-24.
- ^Russ Rowlett. 'Glasgow Coma Scale'. University of North Carolina at Chapel Hill.
- ^Hutchinson’s clinical methods 22nd edition
- ^Iankova, Andriana (2006). 'The Glasgow Coma Scale: clinical application in Emergency Departments'. Emergency Nurse. 14 (8): 30–5. doi:10.7748/en2006.12.14.8.30.c4221. PMID17212177.
- ^'Resources data'(PDF). www.cdc.gov.
- ^Gill M, Windemuth R, Steele R, Green SM (2005). 'A comparison of the Glasgow Coma Scale score to simplified alternative scores for the prediction of traumatic brain injury outcomes'. Ann Emerg Med. 45 (1): 37–42. doi:10.1016/j.annemergmed.2004.07.429. PMID15635308.
- ^Green S. M. (2011). 'Cheerio, Laddie! Bidding Farewell to the Glasgow Coma Scale'. Annals of Emergency Medicine. 58 (5): 427–430. doi:10.1016/j.annemergmed.2011.06.009. PMID21803447.
- ^Iver, VN; Mandrekar, JN; Danielson, RD; Zubkov, AY; Elmer, JL; Wijdicks, EF (2009). 'Validity of the FOUR score coma scale in the medical intensive care unit'. Mayo Clinic Proceedings. 84 (8): 694–701. doi:10.4065/84.8.694. PMC2719522. PMID19648386.
- ^Fischer, M; Rüegg, S; Czaplinski, A; Strohmeier, M; Lehmann, A; Tschan, F; Hunziker, PR; Marschcorresponding, SC (2010). 'Inter-rater reliability of the Full Outline of UnResponsiveness score and the Glasgow Coma Scale in critically ill patients: a prospective observational study'. Critical Care. 14 (2): R-64. doi:10.1186/cc8963. PMC2887186. PMID20398274.
Sources[edit]
- Teasdale G, Murray G, Parker L, Jennett B (1979). 'Adding up the Glasgow Coma Score'. Acta Neurochir Suppl (Wien). 28 (1): 13–6. doi:10.1007/978-3-7091-4088-8_2. ISBN978-3-7091-4090-1. PMID290137.
- Meredith W, Rutledge R, Fakhry SM, Emery S, Kromhout-Schiro S (1998). 'The conundrum of the Glasgow Coma Scale in intubated patients: a linear regression prediction of the Glasgow verbal score from the Glasgow eye and motor scores'. J Trauma. 44 (5): 839–44, discussion 844–5. doi:10.1097/00005373-199805000-00016. PMID9603086.
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Glasgow_Coma_Scale&oldid=900784937'
